Dr. Peter L. Knepell
President of Peak Quality Services
If you want to buy an electronic thermometer, a blood pressure cuff, a pulse-oximeter, or a blood glucose meter, you probably do not give much thought about its accuracy or precision. Should you? More importantly, how about manufacturing these or other medical devices that depend on measurements? Of course, it is important and, often, a safety issue.
The US FDA recognizes this and wants manufacturers to consider Test Method Validation (TMV). US and international standards are published under the title, Measurement System Analysis (MSA). It does not matter what you call it, the big question is: “How do you know you have quality if you cannot measure?”
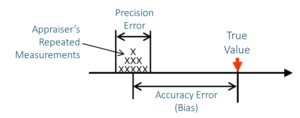
There are some fundamental considerations, like accuracy and precision. Accuracy is the difference between the average of your measurements and the true or expected value. Precision is the dispersion or spread of the readings. This is easy to illustrate.
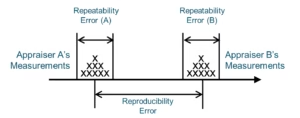
We can break down precision error into two categories: repeatability and reproducibility error. Repeatability is the ability of a person or instrument to repeat measurements. Reproducibility is the ability of two different people or instruments to agree with measurements of the same item. Again, this is easy to illustrate.
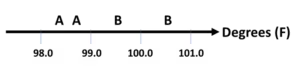
Let’s make this relevant to a typical patient, call him Jim. Suppose Jim goes to a store to buy an electronic thermometer and there are two models available. One requires a swipe on the forehead and the other requires that he just hold the tip six inches from his forehead. Jim asks to try each. He sits down, relaxes, tries the first model and it reads, 98.7° F. Out of curiosity, he turns it off and back on, then tries it again, and it reads 98.4°F. He then tries the second model the same way and it reads 99.5°F and 100.6°F. This simple illustration shows a lot.
So, both repeatability and reproducibility error are easy to see. It looks like Model A is better based on repeatability error. There is reproducibility error but we cannot decide on a better model based on that error. (However, if you are the manufacturer making both models, you have identified a quality problem with your products.) Since we do not know the true value, we cannot comment on accuracy. But, if you ask Jim, he’ll tell you he’s not feeling like he has a fever. So, again, Model A looks better.
Of course, there are more considerations involved with TMV/MSA. For example, how many items and measurements should you involve in your study? Are there certain performance metrics that tell you if your measurement system is acceptable or not? The answers to these questions are given in standards, like ASTM-E2782 or MSA-4 from ISO/TS 16949.
To make it more user-friendly, GBA Key2Compliance is offering a two-webinar series, called
Test Method Validation (TMV) – Made Easy, on Feb 24 and 25, 2026.
For those involved with measurements made by laboratories or diagnostic medical devices, a related webinar on Validation of Analytical Procedures (VAP) will be offered on March 4, 2026.